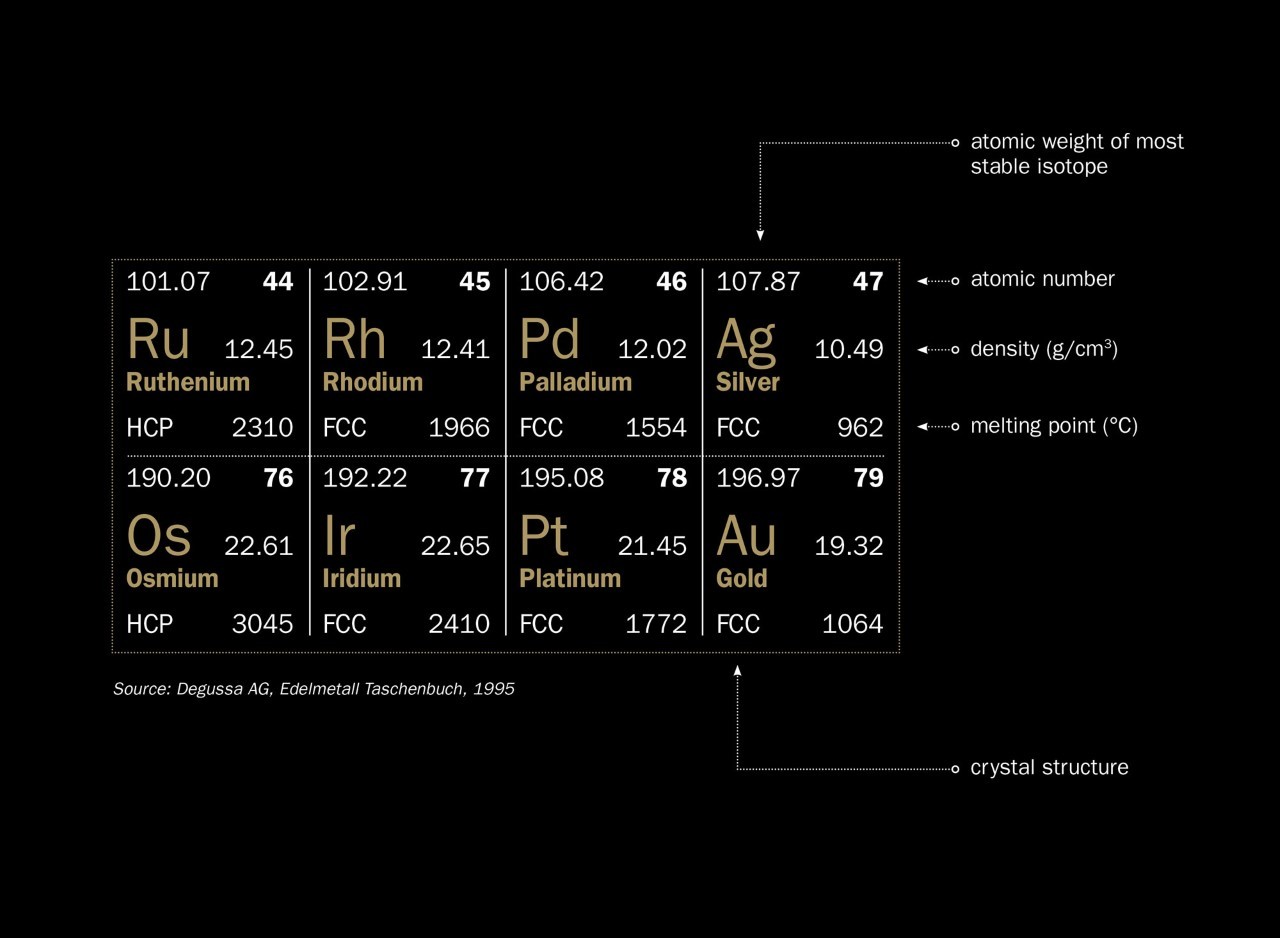
CHAPTER 4 4.4 Calculate the number of vacancies per cubic meter in iron (Fe) at 850°C. The energy for vacancy formation is 1.0
ME 212 Materials Science 11 March 2016 Chapter 6 Problem Set 6.2 Calculate the number of vacancies per cubic meter in iron at 85

Palladium Pd Metal 10mm Density Cube 99.95% Pure For Element Collection - Magnetic Materials - AliExpress
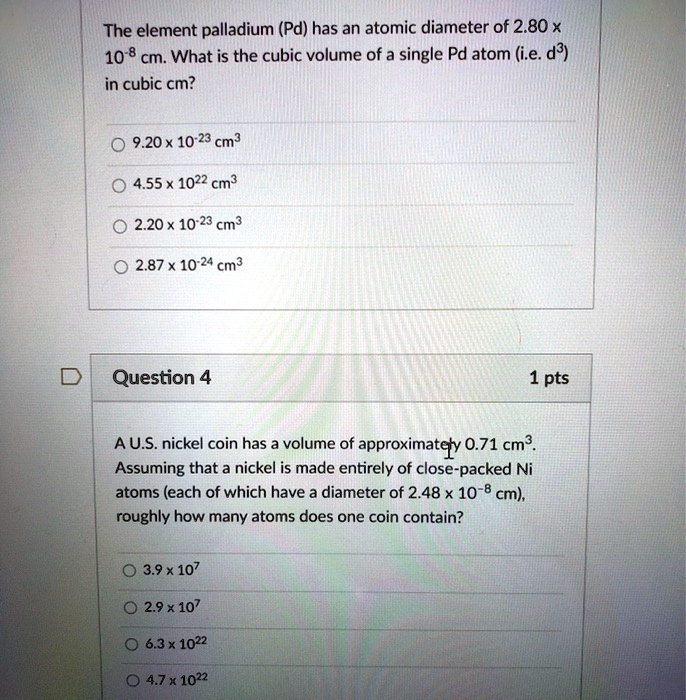
SOLVED: The element palladium (Pd) has an atomic diameter of 2.80 x 10-8 cm: What is the cubic volume of a single Pd atom (i.e: d3) in cubic cm? 9.20x 10-23 cm3

Supported Palladium on Magnetic Nanoparticles–Starch Substrate (Pd-MNPSS): Highly Efficient Magnetic Reusable Catalyst for C–C Coupling Reactions in Water | ACS Sustainable Chemistry & Engineering

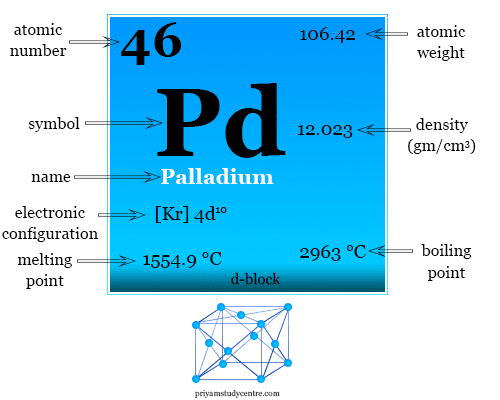
Module-10.pdf - Numerical on vacancy defects The density of a sample of FCC palladium is 11.98 g/cm3 and its lattice parameter is 3.8902 Å. Atomic | Course Hero

fundamentals of materials science and engineering4장 - 35 4 Im per f ec t i ons i n t he At om i c - Studocu
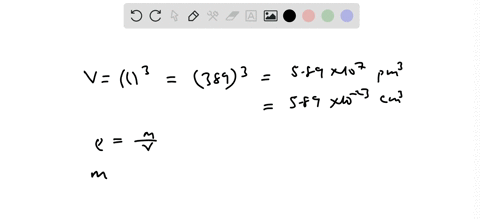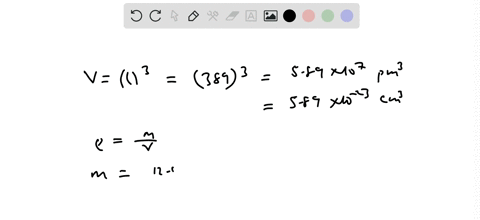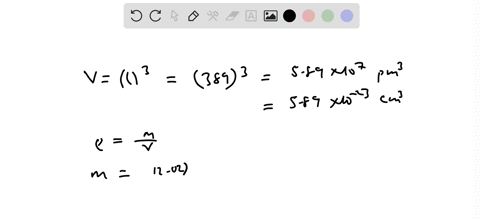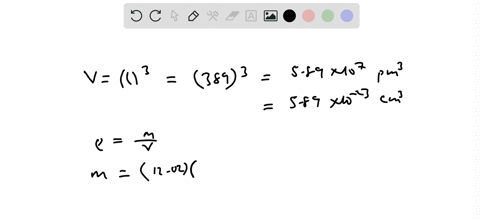
SOLVED:Palladium has a cubic crystal structure in which the edgc of the unit cell is 389 pm long. If the density of palladium is 12.02 g / cm^3, how many palladium atoms

High Purity Metal Palladium, Electrochemical Electrode, Palladium Electrode, Pure Palladium Wire, Purity 99.95+, Diameter 0.6mm, - Ph Meters - AliExpress